Advantages of Lithium Iron Phosphate batteries over Lead-Acid Batteries
Battery storage is an integral part of all energy systems. There are various types of batteries that have been used and the most popular two types at the moment are Lithium Iron Phosphate (LiFePO4) battery and Lead-Acid battery. The LiFePO4 battery uses Lithium Iron Phosphate as the cathode material and a graphitic carbon electrode with a metallic backing as the anode, whereas in the lead-acid battery, the cathode and anode are made of lead-dioxide and metallic lead, respectively, and these two electrodes are separated by an electrolyte of sulfuric acid. The working principle of these two types of batteries is shown in Figures 1(a) and 1(b), respectively.
(a)  (b)
(b)
Figure 1: Working principle of (a) LiFePO4 and (b) lead-acid battery.
In comparison with the lead-acid battery, LiFePO4 poses several advantages that we’ll take a look at in detail in the following sections:
LiFePo4 has Longer Life Cycle: One of the main advantages of lithium iron phosphate batteries is the longer cycle life as compared to lead-acid batteries. On average, LiFePO4 batteries can last for 2,000 to 5,000 charge and discharge cycles without harming their performance. On the other hand, lead-acid batteries range only 200 to 500 cycles. Basically, LiFePO4 batteries have a life span of approximately 5-10 years in comparison with lead-acid batteries that need to be changed every 1-3 years. Comparative analysis as conducted by the researchers established that LiFePO4 batteries have low loss and extended cycle life with lower storage depletion rate as compared to lead-acid batteries regarding the application in the electrical microgrid system [2].
Higher Efficiency: LiFePO4 batteries operate with much lower resistance and can be utilized to approximately 100% of their available capacity. Additionally, these types of batteries are characterized by fast charging and discharging rates, which increases operational efficiency. In comparison to lead-acid batteries, LiFePO4 batteries present 25-35% more efficiency. For example, a lead-acid battery with a capacity of 10Ah will deliver 6.5Ah of charge, whereas a LiFePO4 battery with the same charge capacity delivers almost the full 10Ah. Therefore, a solar system with a specific rating (Ah/Watt) can be designed with 28% less storage capacity. Higher efficient performance of lithium batteries is one of the vital reasons behind its superiority in using as a grid energy storage device [3].
Light Weight: Due to the extremely high energy density, LiFePO4 batteries are relatively compact in size and weight and need less space for installation. This type of battery is about 25-30% of the size and weight of an equivalent lead-acid battery, which is helped by the much higher depth-of-discharge available in a lithium battery. Moreover, LiFePO4 battery systems are generally made up of smaller, easy to handle modules of sizes from 1-2 kWh, which gives much more flexibility in designing a system. The lithium battery finds extensive use in its applications where there are concerns about battery weight and size such as Electric vehicles, RV storage solutions, and off-grid lithium projects. A report states that 750Kg of lead-acid batteries is required to properly power an LDV light van. On the other hand, the same power demand can be met by about 200 Kg of Lithium batteries, resulting in a significant reduction of the mechanical load of the van.
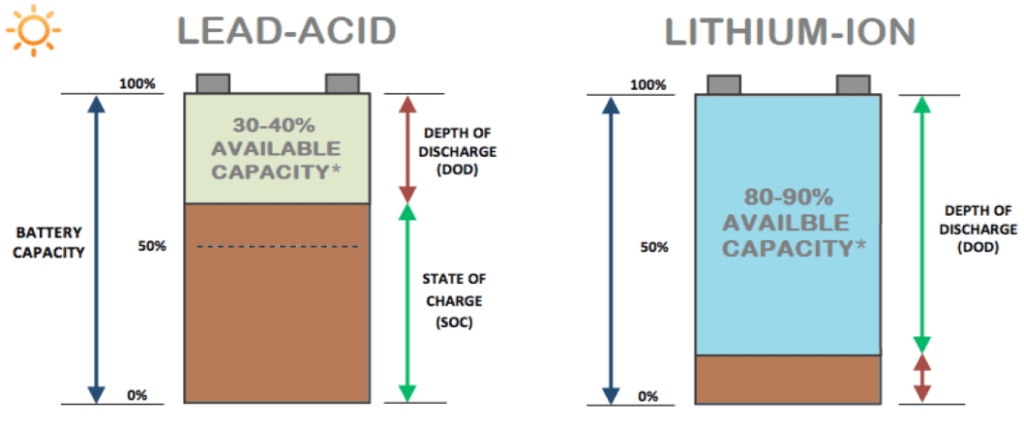
Battery Usable Capacity: LiFePO4 batteries are less susceptible to problems caused by the depth of discharge. As indicated in Figure 2, LiFePO4 batteries are designed to be discharged up to 80-90% of the total capacity without long-term damage, while the traditional lead-acid batteries should not be discharged more than 30-40%. In any emergency situation, lead-acid battery can be discharged to a higher percentage of its capacity but it will result in a reduction in its long-term health. In practical applications, the maximum depth of discharge is about 35% for a lead-acid battery, and a maximum of 60% in backup situations. On the contrary, the LiFePO4 battery can be discharged to 85% on a daily basis, and up to 100% when occasionally needed. Due to the higher battery usable capacity, a system with the same power demand can be designed with less number of LiFePO4 batteries. Researchers conducted a microgrid system design study where they established that utilization of LiFePO4 batteries as a storage medium requires up to 45% less batteries as compared to lead-acid batteries [2].

Higher Working Voltage: LiFePO4 batteries discharge at a constant voltage close to 3.2 V until the cell is completely exhausted. Therefore, the battery can deliver full power until discharged, and it can greatly simplify or even eliminate the need for voltage regulation circuitry. High cell voltage means fewer battery cells, less installation space, and fewer electronics equipment for high-voltage applications. Because of a higher output voltage, four cells are enough to generate 12.8 V of DC. On the contrary, to generate the same voltage level, 5-6 cell of lead-acid batteries, each having a capacity of 2.30-2.35V is required to be connected in series. A higher cell voltage makes LiFePO4 batteries a practical replacement for lead-acid batteries in the automotive and solar applications.
Lesser Maintenance: And, as if there weren't already enough advantages of lithium iron phosphate batteries over lead-acid, they also don’t require active maintenance to extend their service life. Since these batteries have no memory effects and a much smaller self-discharge rate, they can be stored for a longer period of time with almost no ill-effect. On the other hand, lead-acid batteries need special maintenance, or you risk reduction of their overall life span.
Safety: From the standpoint of safety measurements, LiFePO4 batteries are more thermally and chemically stable as compared to lead-acid batteries because of the absence of liquid electrolyte that is more prone to leaking [1]. LiFePO4 is an intrinsically safer cathode material and LiFePO4 cells are harder to ignite in the event of charging. Moreover, they do not decompose at high temperatures. The bad rap Lithium batteries have, stems from the original Li-ion (Cobalt based). They were and still are not nearly as safe as LiFePO4 batteries.
Higher Rate of Charging and Discharging: In comparison with the lead-acid battery, the LiFePO4 battery offers a higher charging and discharging rate [5]. A higher charging rate causes less time to fully charge the battery, whereas a higher discharging rate causes a high output current. As a result, this battery is becoming preferable for electric-powered automotive industries that need a higher acceleration in the beginning. Typically, you can expect a 1C discharge rate from LiFePO4 batteries. That means if the capacity “C” is 100Ah on a LiFePO4 battery, then you can typically safely charge and discharge it at 100 Amps (check your specific batteries specifications). There is also no multi-stage charging with LiFePO4 batteries, which means you can apply the max number of amps into the battery until it hits 100%. Whereas with lead-acid, you must step the amps down as the state of charge increases through stages. These stages are known as Bulk, Absorption, and Float in lead-acid batteries. The stages cause lead-acids to need considerably more time to reach 100% state of charge compared to LiFePO4 batteries.
Wider Temperature Range: LiFePO4 batteries can safely operate over a wide temperature range, typically from –20°C to 60°C, which makes them a good fit for all-weather conditions including extreme cold temperature. In fact, this type of battery offers better performance at colder temperatures than the lead-acid battery. For example, at 0°C, a lead-acid battery’s capacity is reduced by up to 50%, while a LiFePO4 battery suffers only a 10% loss [6]. There have been numerous studies that show lead-acid batteries have drastically reduced, as much as 90% in cold weather under heavy power loads.
Environmental Friendliness: The main components of lead-acid batteries are lead and sulfuric acid, which contaminate the environment and damage the ecosystem if improperly discarded. On the other hand, the electrodes of the LiFePO4 are made of non-toxic materials, which can also be recycled to recover the materials. From an environmental standpoint, LiFePO4 batteries pose far less risk to the environment than lead-acid batteries. We have a great article on where lithium comes from and what makes it special if you'd like to learn more about it.
Wrap Up: As you can see, the facts show why we always recommend LiFePO4 batteries as a better solution to lead-acid batteries across almost all applications. The main problems of utilizing LiFePO4 batteries are their availability and 3-5 times higher cost as compared to a similarly sized lead-acid battery. However, over recent years, with the increase in popularity and technological advancement, the price of LiFePO4 batteries continues to fall. From a total cost of ownership standpoint, LiFePO4 batteries are much cheaper if you take into account the number of times you’ll replace lead acid (5-10) to equal the life span of just one Lithium Iron Phosphate battery.
References
[1] "Rechargeable Lithium Batteries". Electropaedia — Battery and Energy Technologies.
[2] S. Dhundhara, Y.P. Verma, A. Williams, “Techno-economic analysis of the lithium-ion and lead-acid battery in microgrid systems”, Energy Conversion and Management, vol. 177, pp. 122-142, 2018.
[3] T. Chen, Y. Jin, H. Lv, A. Yang, M. Liu, B. Chen, Y. Xie and Q. Chen, “Applications of Lithium-Ion Batteries in Grid-Scale Energy Storage Systems” Transactions of Tianjin University Springer, vol. 26, pp. 208–217, 2020.
[4] https://www.cleanenergyreviews.info/blog/simpliphi-pylontech-narada-bae-lead-acid-battery
[5] M. Egashira, A. Kanetomo, N. Yoshimoto, M. Morita, “Charge–discharge rate of spinel lithium manganese oxide and olivine lithium iron phosphate in ionic liquid-based electrolytes”, Journal of Power Sources, vol. 196, issue 15, Aug 2011, pp. 6419-6424.
[6] D. Lu, S. Lin, W. Cui, S. Hu, Z. Zhang, W. Peng, “Swelling mechanism of 0%SOC lithium iron phosphate battery at high temperature storage”, Journal of Energy Storage, vol. 32, Dec. 2020, 101791.