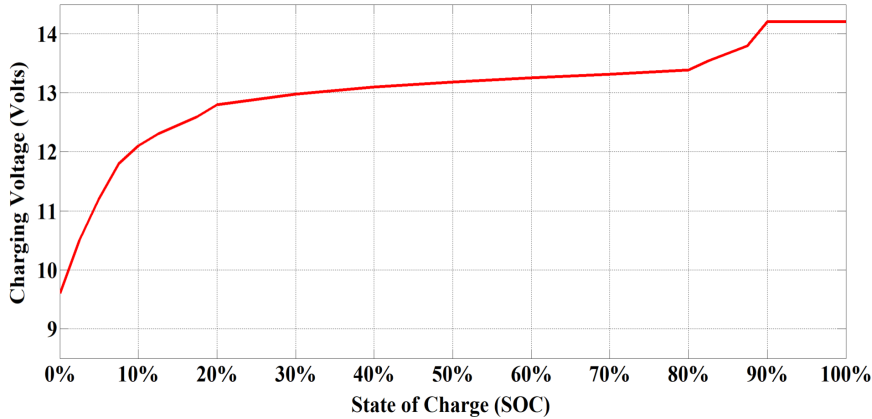
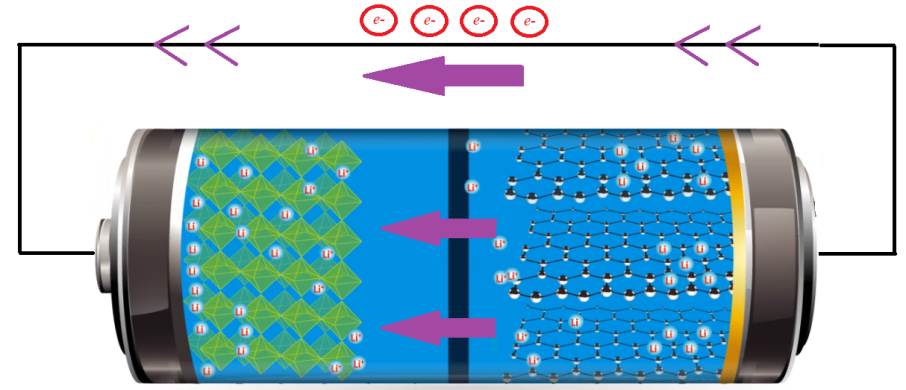
In comparison with the conventional lead-acid battery, lithium iron phosphate (LiFePO4) batteries offer several advantages such as increased life cycles, improved discharge efficiency, lighter weight, climate resistance, and the ability to deep cycle [1]. Although LiFePO4 batteries are often more expensive than lead-acid batteries the minimal maintenance and infrequent replacement make them a valuable investment and smart long-term solution. As a consequence, the LiFePO4 batteries are gaining popularity day by day for many domestic and industrial applications. In the following section, different applications of LiFePO4 batteries and the reason behind their suitability over other types of batteries are analyzed in detail.
Electric Vehicles: Under the circumstances of growing environmental concern, most progressive countries are in the process to increase the usage of electric-powered vehicles to reduce emissions. The most integral part of the electric vehicle is the battery that acts as an energy source to drive the electric motor. In recent days, lead-acid batteries are being replaced with LiFePO4 batteries for electric vehicle application, which is because of the tremendous advantages of LiFePO4 batteries such as fast charging and discharging rate, wider temperature range, and longer cycle life. It is obvious that to match up with the standard of internal combustion (IC) engine, the electric cars must have a higher acceleration at the beginning that can be perfectively delivered by the LiFePO4 battery as it has a high discharge rate and accordingly, a higher output current [2]. On the other hand, a higher charging rate compared to lead-acid battery makes it easier for electric vehicle owners to charge the vehicle in a shorter time. Moreover, the performance of the LiFePO4 battery does not deteriorate much with the variation in temperature. For example, at 0°C, a lead-acid battery’s capacity is reduced by up to 50%, while a LiFePO4 battery suffers only a 10% loss [3]. Therefore, LiFePO4 batteries are the best choice in an extremely cold or hot region.
Energy Storage: Utilization of the energy storage system is essential for powering the electrical loads, especially where there is no/less access to electricity. For example, a local microgrid system having a higher percentage of intermittent renewable energy sources must utilize an energy storage system to meet the load demand at each instant. In this regard, LiFePO4 batteries when combined with converters and inverters can perform better in comparison with other types of batteries because of higher usable capacity and higher efficiency. LiFePO4 batteries are designed to be discharged up to 80-90% of the total capacity without long-term damage, while the traditional lead-acid batteries should not be discharged more than 30-40%. In any emergency situation, a lead-acid battery can be discharged to a higher of its capacity but it will cause a reduction in its cycle life. Due to the higher battery usable capacity, a system with the same power demand can be designed with less number of LiFePO4 batteries. Researchers conducted a microgrid system design study where they established that utilization of LiFePO4 batteries as storage medium requires up to 45% lesser batteries as compared to lead-acid batteries [1]. On the other hand, LiFePO4 batteries operate with much lower resistance and can be utilized to approximately 100% of their available capacity. Compared to lead-acid batteries, LiFePO4 batteries present 25-35% more efficiency making them superior in use as a grid energy storage device [4].
Solar Photovoltaic System: Solar photovoltaic is a renewable energy source that converts sunlight into electrical energy. The generated electric energy can be utilized to meet the load demand during the daytime, but, during the night time, there will be no generation of the solar system. Therefore, the excess energy during the day time can be stored in the battery storage system to further use at night time. In this regard, LiFePO4 batteries can be used with solar charge controllers as energy storage elements in the solar system [5]. Compared to lead-acid batteries, LiFePO4 batteries are advantageous in solar application mainly due to outstanding cycle life. On average, LiFePO4 batteries can last for 1,000 to 3,000 charge and discharge cycles without deferring their performance. On the other hand, lead-acid batteries range only 200 to 1000 cycles. Basically, LiFePO4 batteries have a life span of approximately 5-10 years in comparison with lead-acid batteries that need to be changed every 1-3 years. Comparative analysis as conducted by the researchers established that LiFePO4 batteries have low loss and extended cycle life with a lower storage depletion rate as compared to lead-acid batteries [1]. Moreover, LiFePO4 batteries do not require as much maintenance and replacements as compared to lead-acid batteries and these batteries are also more environmentally friendly.
Boats and Light Van: Batteries must be utilized in boats and light vans for providing electrical power. In such a context, LiFePo4 batteries offer tremendous advantages over other types of batteries since LiFePO4 batteries are less in weight and have a longer life cycle. Due to the extremely high energy density, LiFePO4 batteries are relatively compact in size and weight and need less space for installation. This type of battery is about 25% of the size and weight of an equivalent lead-acid battery, which is helped by the much higher depth-of-discharge. LiFePo4 batteries find their application when there are concerns about battery weight and size such as boats and Light Vans. A report states that 750Kg of lead-acid batteries is required to properly power a light van. On the other hand, the same power demand can be met by about 200Kg of LiFePo4 batteries, resulting in a significant reduction of the mechanical load. Moreover, LiFePO4 batteries have a higher life cycle, lower loss, and lower storage depletion rates as compared to lead-acid batteries.
Communication Equipment: In the context of communication technology, LiFePO4 batteries are gradually replacing other types of batteries to be used as radio communications equipment due to their longevity, lightweight, safety profile, and less maintenance. In comparison to lead-acid batteries, the weight and size of LiFePO4 batteries are significantly less, making this battery most preferable for a communication system that demands less space for installation. Moreover, LiFePO4 battery systems are generally made up of smaller, easy to handle modules of sizes from 2-4 kWh, which gives much more flexibility in designing a low voltage communication system. LiFePO4 batteries are also safe and chemically stable. Therefore, in case of hazardous events, such as collision or short-circuiting, they won’t explode or catch fire, significantly reducing any chance of harm.
Apart from the major applications as described, the safe and reliable factor and the environmental friendliness make LiFePO4 batteries versatile for powering and charging general-purpose and instrumentation equipment such as computers, amplifiers, mobile phones, scissor lifts, electric green mowers, and various types of sensors.
References
[1] S. Dhundhara, Y.P. Verma, A. Williams, “Techno-economic analysis of the lithium-ion and lead-acid battery in microgrid systems”, Energy Conversion and Management, vol. 177, pp. 122-142, 2018.
[2] M. Egashira, A. Kanetomo, N. Yoshimoto, M. Morita, “Charge–discharge rate of spinel lithium manganese oxide and olivine lithium iron phosphate in ionic liquid-based electrolytes”, Journal of Power Sources, vol. 196, issue 15, Aug 2011, pp. 6419-6424.
[3] D. Lu, S. Lin, W. Cui, S. Hu, Z. Zhang, W. Peng, “Swelling mechanism of 0%SOC lithium iron phosphate battery at high-temperature storage”, Journal of Energy Storage, vol. 32, Dec. 2020, 101791.
[4] T. Chen, Y. Jin, H. Lv, A. Yang, M. Liu, B. Chen, Y. Xie, and Q. Chen, “Applications of Lithium-Ion Batteries in Grid-Scale Energy Storage Systems” Transactions of Tianjin University Springer, vol. 26, pp. 208–217, 2020.
[5] X. Wanga, P. Adelmann, T. Reindl, “Use of LiFePO4 Batteries in Stand-Alone Solar System”, Energy Procedia, vol. 25, 2012, pp. 135 – 140.